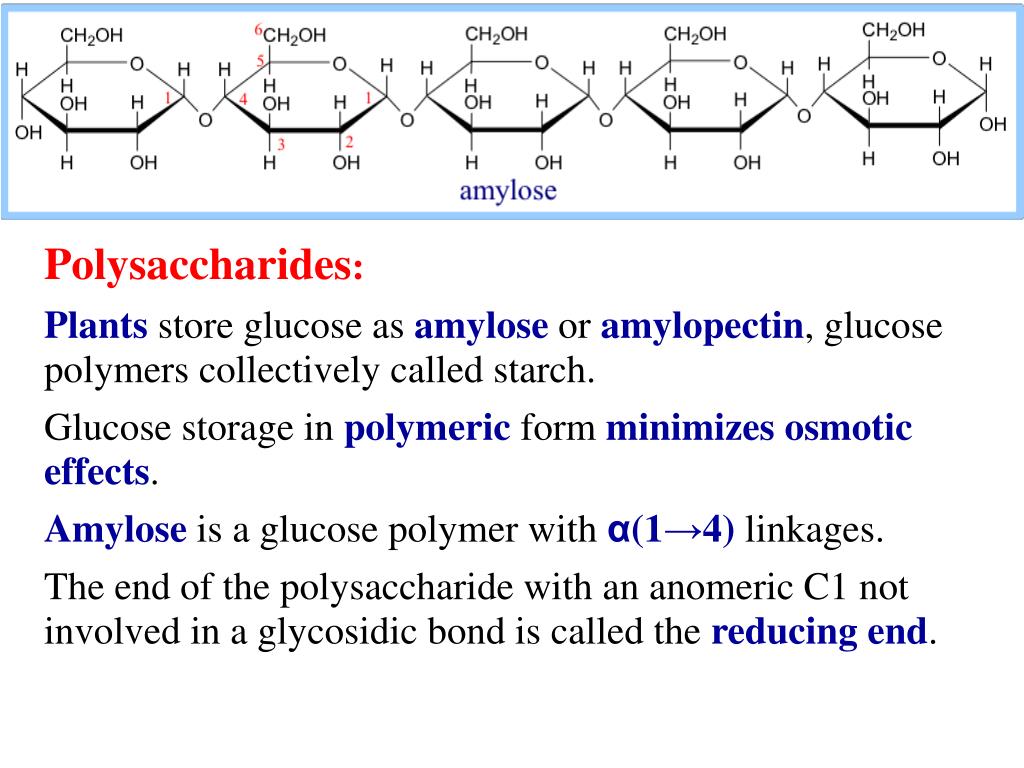
Glycogen is the storage form of glucose in humans and other vertebrates. The starch in the seeds provides food for the embryo as it germinates while the starch that is consumed by humans is broken down by enzymes into smaller molecules, such as maltose and glucose. Starch is the stored form of sugars in plants and is made up of glucose monomers that are joined by α1-4 or 1-6 glycosidic bonds. Plants are able to synthesize glucose, and the excess glucose is stored as starch in different plant parts, including roots and seeds. Starch, glycogen, cellulose, and chitin are primary examples of polysaccharides. The chain may be branched or unbranched, and it may contain different types of monosaccharides. 
PolysaccharidesĪ long chain of monosaccharides linked by glycosidic bonds is known as a polysaccharide (poly- = “many”). The most common disaccharide is sucrose, or table sugar, which is composed of the monomers glucose and fructose. Maltose, or malt sugar, is a disaccharide formed by a dehydration reaction between two glucose molecules. Lactose is a disaccharide consisting of the monomers glucose and galactose. In sucrose, a glycosidic linkage is formed between carbon 1 in glucose and carbon 2 in fructose.Ĭommon disaccharides include lactose, maltose, and sucrose. By convention, the carbon atoms in a monosaccharide are numbered from the terminal carbon closest to the carbonyl group. In the process, a water molecule is lost. Common Disaccharides Disaccharides: Sucrose is formed when a monomer of glucose and a monomer of fructose are joined in a dehydration reaction to form a glycosidic bond. Glycosidic bonds (also called glycosidic linkages) can be of the alpha or the beta type. A covalent bond formed between a carbohydrate molecule and another molecule (in this case, between two monosaccharides) is known as a glycosidic bond. During this process, the hydroxyl group of one monosaccharide combines with the hydrogen of another monosaccharide, releasing a molecule of water and forming a covalent bond. Disaccharidesĭisaccharides (di- = “two”) form when two monosaccharides undergo a dehydration reaction (also known as a condensation reaction or dehydration synthesis). Glucose and galactose are aldoses, and fructose is a ketose. This makes them different molecules despite sharing the same atoms in the same proportions, and they are all isomers of one another, or isomeric monosaccharides. dehydration reaction: A chemical reaction in which two molecules are covalently linked in a reaction that generates \text_6), they differ structurally and stereochemically.isomer: Any of two or more compounds with the same molecular formula but with different structure.A polysaccharide is a long chain of monosaccharides linked by glycosidic bonds the chain may be branched or unbranched and can contain many types of monosaccharides.Sucrose (table sugar) is the most common disaccharide, which is composed of the monomers glucose and fructose.Disaccharides form when two monosaccharides undergo a dehydration reaction (a condensation reaction) they are held together by a covalent bond.Glucose, galactose, and fructose are monosaccharide isomers, which means they all have the same chemical formula but differ structurally and chemically.Monosaccharides are simple sugars made up of three to seven carbons, and they can exist as a linear chain or as ring-shaped molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
